Care for underserved and marginalized populations happens more in community cancer centers than anywhere else—yet these centers and the populations they serve remain significantly underrepresented in clinical research.1 Historically, access to clinical trials has been limited in these settings due to insufficient infrastructure, limited staffing, challenges with patient recruitment, lack of training, and other constraints.1
In 2022, the Association of Cancer Care Centers (ACCC) recognized this gap and sought to address it through an initiative to expand clinical trial access in community oncology settings. Through a comprehensive member survey and analysis of US census data, a national heat map was developed, pinpointing clinical trial activity across the country and highlighting areas of opportunity and underrepresentation.
Building on these findings, ACCC—in collaboration with the Association of Clinical Research Professionals (ACRP)— launched a pilot program in 2024 to explore the impact of providing foundational, oncology-specific clinical research training and tools to build research capacity and strengthen clinical trial involvement.
Focusing on self-identified research-naive and low-trialcapacity cancer programs with regionally, racially, and ethnically diverse catchment areas, this program aimed to equip these centers with the knowledge, tools, and mentorship needed to initiate and successfully manage clinical trials.
Piloting a Mentorship Model to Build Research Capacity
Access to clinical trials has been directly linked to improved cancer outcomes and reduced mortality, making trial availability a critical factor in narrowing disparities in cancer care, especially between urban and rural communities.2 However, many barriers persist, including the underrepresentation of racially and ethnically diverse populations. Studies have shown that this underrepresentation is often rooted in systemic issues such as implicit bias, socioeconomic barriers, and simply not being invited to participate.3
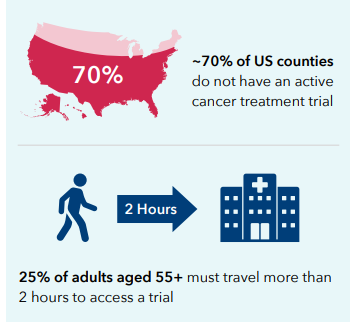
Approximately 70% of US counties lack an active cancer treatment trial, and 25% of adults over age 55 must travel more than 2 hours to access a trial.4
After collaborating with ACRP to establish the foundation for the pilot—including the development of an oncology-specific clinical research training module created by a panel of ACRP oncology subject-matter experts—ACCC led efforts to identify community oncology sites interested in initiating or expanding their clinical research programs. With these challenges in mind, pilot site selection was deliberate and strategic. Participating cancer programs had to meet the following criteria:
- Self-identify as research-naive or having low trial capacity.
- Serve a geographically, racially, ethnically, or financially diverse catchment area.
- Demonstrate institutional support for growing trial capacity.
- Commit to participation in an 8-week training course.
Following a comprehensive interview and selection process, 9 cancer programs representing a cross-section of urban, suburban, and rural areas across the US were chosen. These included:
- Anita Stewart Oncology Center, Tifton Regional Cancer Center in Tifton, Georgia
- Beth Israel Deaconess Plymouth Cancer Center in Plymouth, Massachusetts
- Grand View Health in Sellersville, Pennsylvania
- MercyOne Genesis Cancer Center in Davenport, Iowa
- Prisma Health Cancer Institute–Columbia in Columbia, South Carolina
- Skagit Regional Health Cancer Center in Mount Vernon, Washington
- Southcoast Centers for Cancer Care in Fairhaven, Massachusetts
- Touro Infirmary–LCMC Health in New Orleans, Louisiana
- Woman’s Cancer Pavilion/Woman’s Hospital in Baton Rouge, Louisiana.
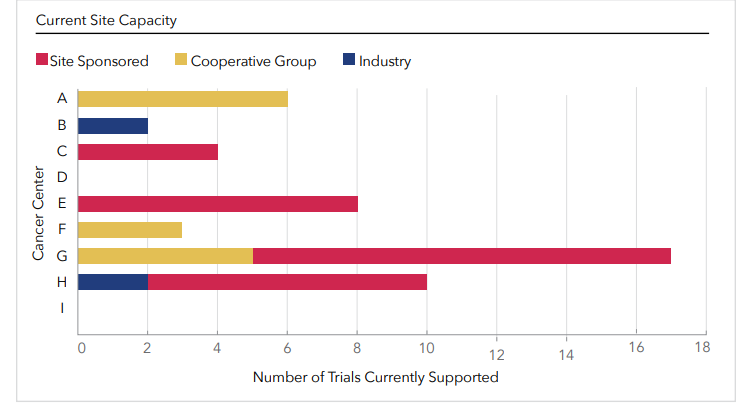
Figure 1. Clinical trial capacity across cancer centers.
At the time of enrollment, each site had 10 or fewer open clinical trials, as shown in Figure 1 (including mixed portfolios of cooperative group trials, investigator-initiated trials, and very few active industry trials). Research teams were small, between 1 to 2 full-time equivalent staff dedicated to oncology research. Among the 16 learners from these sites—including clinical research coordinators, clinical research nurses, an oncology research coordinator, a cancer center administrator, an oncology social worker, and an investigational drug service pharmacist—just under half had ever worked on a clinical trial. Most participants had limited research experience, typically around 2 years, though 1 participant had significantly more, with 40 years of experience.
To support their development, selected sites received access to a bevy of tools and resources as part of the training:
- Free enrollment in an oncology-specific clinical trials training program (ACRP Clinical Research Foundations course with an Introduction to Oncology Clinical Trial Nuances)
- Opportunities for peer-to-peer learning and participation in virtual collaboration sessions
- Access to ACCC’s Community Oncology Research Institute (ACORI) clinical trial tools, including a site self-assessment, implicit bias training, and clinical trials glossary
- Guided mentorship training through site feasibility and trial enrollment.
An Inside Look at the Training
The program was designed to provide flexible, accessible education for busy staff while building core competencies in oncology-specific clinical research. A blend of self-paced learning with live, expert-led virtual sessions enabled learners to balance training with daily responsibilities.
The self-study curriculum covered foundational topics such as clinical trials, ethical and regulatory considerations, statistical guidelines, and global standards. Live virtual sessions examined real-world challenges, such as site feasibility, trial monitoring, safety reporting, investigator responsibilities, and patient recruitment and retention strategies.
To complement this, participants also engaged with ACORI’s suite of equity-focused tools, including the Just AskTM Increasing Diversity in Cancer Clinical Research online training program and site self-assessment. These resources helped learners identify implicit and conscious bias and offered actionable strategies to reduce disparities in cancer research settings.
Measuring Impact: Learner Insights
To understand the effectiveness of the training program, ACCC worked with ACRP to evaluate participants’ experiences across the pilot sites through pre- and post-intervention self-reported assessments.
Overall, participants reported that the training enhanced their knowledge and understanding of clinical trial operations and workflows—particularly key roles and responsibilities involved in running a study—and inspired them to advocate for future research opportunities at their institutions. One participant shared, “The oncology training program was excellent, and I learned a lot.” Several attendees also expressed interest in pursuing additional learning, especially training that could lead to professional certification.
Learners reported increased understanding of methods to increase participation of diverse patient populations in trials, as well as improved knowledge of diversity, equity, and inclusion principles in research.
"We are patient-centered in our clinical trials awareness-building approach and look forward to educating our patients about opportunities to participate in clinical trials. Our approach is to focus on the patient’s experience and to ensure that the consenting process for trials is truly that—a process that spans the life of their participation. We check in with our patient volunteer to ensure they understand their participation and provide information to answer questions and concerns at every encounter with our team. We do not want clinical trials participation to be a burden. Rather, we ensure that patients feel heard regarding all aspects of their participation and that they know that they are the most important stakeholder at the center of our attention as we conduct clinical trials in our community.”
— Kemi Oladipupo, MSHS, MPH, BSN, RN, CCRP
Clinical Research Coordinator Touro Cancer Center
Touro Cancer Center
The format—a blend of self-paced learning with live, instructorled sessions—was well-received. Participants found the instruction engaging and appreciated the flexibility to learn at their own pace. Still, many noted the challenge of balancing the training with their regular day-to-day job responsibilities, suggesting that future programs should consider ways to provide protected time for participants. “I was able to carve time out for the training; if I had to see a patient, I would drop off briefly and come back,” noted one participant, highlighting the competing demands many faced.
Notably, the self-study materials stood out as the most helpful aspect of the program. Learners found them relevant, easy to navigate, and instrumental in preparing for live sessions.
Looking ahead, participants recommended building on this foundation with more advanced content. Suggestions included deeper dives into FDA regulations, mock site audits, and modules tailored to specific professional roles—such as pharmacy and social work—to better address the multidisciplinary nature of clinical trial teams.
Lessons Learned to Build Sustainable Research Capacity
As ACCC continues to evaluate the program’s impact, several practical takeaways have emerged that can inform future efforts to build research capacity within community cancer centers, particularly those operating with limited staffing or insufficient clinical trial exposure.
Customized Training to Participants’ Level of Research Experience
Because most learners came from diverse professional backgrounds with varying levels of familiarity with clinical research, a one-size-fits-all approach presented challenges during the pilot. Incorporating a preassessment of knowledge levels and potentially offering distinct learning tracks for foundational and intermediate learners could enhance the training experience.
Protected Time and Institutional Buy-In
Balancing the demands of training alongside clinical responsibilities proved difficult for many participants, especially as they were the only research staff at their center. Future iterations of this model would benefit from institutional support that ensures dedicated time for staff to fully engage with the training and collaborative learning opportunities.
Mentorship and Community-Building
Beyond learning the mechanics of conducting clinical trials, participants found great value in the opportunities to connect with peers at similarly resourced sites. The ability to share insights, discuss challenges unique to low-capacity or research-naive settings, and exchange resources helped foster a sense of community that could support continued engagement after the completion of training.
More Oncology-Specific Content
Learners expressed the desire for more education centered around oncology. The nuances of oncology clinical trials—from differences in funding, workflow, and regulatory oversight— present an opportunity for greater guidance on how to move from readiness to launching live trials. Expanded content in these areas could help bridge the gap between theoretical knowledge and real-world application.
“We are always open to opportunities to enhance our knowledge and understanding of the best practices for the quality conduct of clinical trials, and that is quality as it pertains specifically to patient safety and data integrity. The opportunities for learning that ACCC has provided our team has exposed us to new knowledge about ICH-CGP E6 (R3), and insights shared on this updated guidance for industry has given us some critical tools for refining our SOPs for data management in clinical trials.”
— Kemi Oladipupo, MSHS, MPH, BSN, RN, CCRP
Clinical Research Coordinator
Touro Cancer Center
Looking Ahead: Investing in Community-Based Research
This pilot program marks a pivotal step in the ongoing effort to increase clinical trial participation at community cancer centers. The insights that learners gained highlight the untapped potential of research-naive sites to meaningfully contribute to clinical research.
By delivering oncology-specific training, practical tools, and mentorship, the initiative demonstrates that community-based programs can build the confidence and competencies needed to initiate and sustain clinical trials. Moreover, these centers, often serving more diverse and underserved patient populations, are uniquely positioned to help advance equitable access to cutting-edge cancer care.
While challenges remain—from limited staffing to infrastructure gaps—the results are encouraging. The lessons learned from this pilot can serve as a blueprint for future efforts. With continued investment in capacity-building and strategic support, community cancer programs can become integral players in oncology research, helping to shape a more inclusive, accessible future for clinical trials. As the health care system continues to prioritize equitable access to care, programs like this pilot are not just helpful—they are essential.
Acknowledgements
ACCC is grateful to the Association of Clinical Research Professionals, the ACORI Advisory Committee, pilot sites, and others who graciously contributed their knowledge and time to this education program.
References
1. Ebrahimi H, Megally S, Plotkin E, et al. Barriers to clinical trial implementation among community care centers. JAMA Netw Open. 2024;7(4):e248739. doi:10.1001/ jamanetworkopen.2024.8739
2. Baranda JC, Diaz FJ, Rubinstein L, et al. Expanding access to early phase trials: the CATCH-UP.2020 experience. JNCI Cancer Spectr. 2023;7(1):pkac087. doi:10.1093/jncics/pkac087
3. Acuña-Villaorduña A, Baranda JC, Boehmer J, Fashoyin-Aje L, Gore SD. Equitable access to clinical trials: how do we achieve it? Am Soc Clin Oncol Educ Book. 2023;43:e389838. doi:10.1200/EDBK_389838
4. ASCO. Cancer clinical trials continue to be out of reach for many patients. Ascopost.com. Accessed April 10, 2025. https:// ascopost.com/issues/october-25-2024/cancer-clinical-trialscontinue-to-be-out-of-reach-for-many-patients