The Centers for Medicare & Medicaid Services (CMS) released its Medicare Physician Fee Schedule (MPFS) on July 7, 2022. Stakeholders have until September 6, 2022, to submit their comments to the agency on the proposed changes for calendar year (CY) 2023. Below are several of the key items in the MPFS proposed rule that relate or impact oncology programs and providers. Note: some payment impacts are outside CMS’ authority to change.
MPFS Conversion Factor
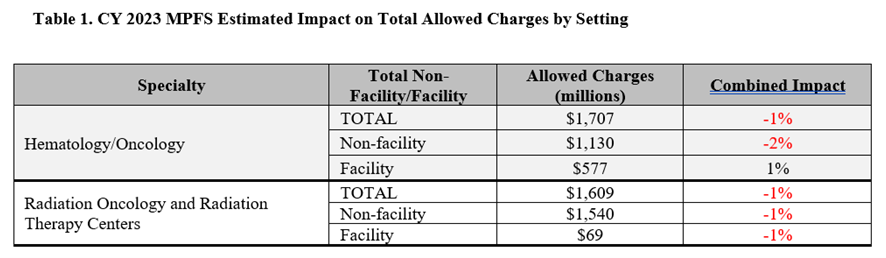
The conversion factor is the value multiplied to the assigned relative value units (RVUs) of physician work, practice expense, and malpractice of each code to determine the dollar amount of each code’s payment. CMS proposed a conversion factor of $33.0775—a decrease of 4.5 percent from the CY 2022 conversion factor of $34.6062. In the table below, CMS provided a breakdown of the proposed payment impacts to oncology specialties by setting. This breakdown only reflects the impact to the estimated RVUs and does not reflect other changes, such as the 4.5 percent decrease to the conversion factor.
CMS also proposed updates to malpractice RVUs for next year; these were last updated in CY 2020 and are required to be updated every three years. Based on the malpractice or practice liability insurance data collected from all 50 states, CMS proposed changes to the risk index values that are used to calculate the malpractice RVUs at the code level. Malpractice RVUs reflect the risk of the primary specialty assigned to the service and that performs the service. For CY 2023, the risk index value for Hematology/Oncology is proposed to decrease from 0.765 to 0.741 for years 2023 to 2025, and, for Radiation Oncology, CMS proposed an increase from 0.840 to 0.905 for years 2023 to 2025.
Evaluation and Management Changes
Effective January 1, 2023, there will be updates to the next set of evaluation and management (E/M) codes. These codes are the “Other E/M” visits (inpatient and observation visits, emergency department [ED] visits, nursing facility visits, domiciliary or rest home visits, home visits, and cognitive impairment assessments). These codes exclude critical care services, yet they match the framework (e.g., medical decision making or time-base) of the outpatient and office E/M visits that changed in 2021. CMS proposed to accept and move forward with the American Medical Association (AMA) Current Procedural Terminology (CPT®) Editorial Panel changes, with a few minor exceptions. Changes to the “Other E/M” visit codes made by the AMA were released in early July 2022.
CMS clarified its proposal to slightly amend the definitions of “initial” and “subsequent” in relation to E/M visits for inpatient services. The agency does not recognize subspecialties, as is outlined in the CPT manual, so it proposed the following language:
- “An initial service would be defined as one that occurs when the patient has not received any professional services from the physician or other qualified health care professional or another physician or other qualified health care professional of the same specialty who belongs to the same group practice during the stay.”
- “A subsequent service would be defined as one that occurs when the patient has received any professional services from the physician or other qualified health care professional or another physician or other qualified health care professional of the same specialty who belongs to the same group practice during the stay.”
CMS also proposed three new Healthcare Common Procedure Coding System (HCPCS) codes to be used in place of the AMA created CPT code 993X0 for prolonged services. One code would be for hospital inpatient or observation care, another for nursing facilities, and another for home or residence. Starting in 2023, providers should use the below code to bill prolonged services for inpatient time-based visits with their Medicare beneficiaries:
- GXXX1. Prolonged hospital inpatient or observation care evaluation and management service(s) beyond the total time for the primary service (when the primary service has been selected using time on the date of the primary service); each additional 15 minutes by the physician or qualified healthcare professional, with or without direct patient contact (list separately in addition to CPT codes 99223, 99233, and 99236 for hospital inpatient or observation care evaluation and management services). Do not report GXXX1 on the same date of service as any other prolonged service for evaluation and management (99358, 99359, 993X0). Do not report GXXX1 for any time unit less than 15 minutes.
These new HCPCS codes would replace the existing CPT codes for inpatient prolonged services:
- 99356. Prolonged service in the inpatient or observation setting, requiring unit/floor time beyond the usual service; first hour. (List separately in addition to the code for inpatient or observation E/M service.)
- 99357. Prolonged service in the inpatient or observation setting, requiring unit/floor time beyond the usual service; each additional 30 minutes. (List separately in addition to code for prolonged service.)
As with outpatient prolonged services, CMS did not agree with the AMA on how time is counted time to meet the threshold for billing new codes. In addition, the prolonged service code GXXX1 can only be used with the highest-level hospital inpatient or observation care visit codes (99223, 99233, and 99236) when the time-based method is used.
CMS proposed that the prolonged service period described by GXXX1 begins 15 minutes after the total times (as established in the physician time file) for codes 99223, 99233, and 99236 have been met. Additionally, CMS proposed that the GXXX1 prolonged code would be for a 15-minute increment, and the entire 15-minute increment must be completed to bill the code.
CMS also proposed that GXXX1 would apply to face-to-face and non-face-to-face time spent on patient care within the survey timeframe. For codes 99223 and 99233, this would be time spent on the date of the patient encounter. For code 99236, this would be time spent within 3 calendar days of the patient encounter.
CMS proposed to fully integrate split (or shared) E/M visits for new and established patients in 2024 (a one-year delay) to allow full acquaintance and implementation of the other E/M visit changes for healthcare providers.
Telehealth Post-PHE
In the absence of any further extensions or changes to Medicare telehealth policy by Congress, the provisions and waivers that were implemented in response to the COVID-19 pandemic will continue for 151 days after the end of the public health emergency (PHE). Currently, the PHE is scheduled to end on October 13, 2022. CMS reiterated that if any codes are not included in the telehealth list of services that are identified as continuing permanently or temporarily (as a Category 3 telehealth service), they will end on day 152 after the PHE’s end date. Specific to oncology, services to be removed include 77427 for radiation oncology physician management; initial inpatient E/M codes 99221, 99222, and 99223; as well as audio-only codes 99441, 99442, and 99443.
With some exceptions, billing for telehealth services will return to pre-PHE guidelines and will no longer require the use of modifier 95. Instead, the appropriate place of service (POS) code (02 or 10) must be applied to process payment.
Another change is that telehealth visits will no longer be allowed for patients in their homes or anywhere outside of an originating site other than the statutory exceptions for diagnosis, evaluation, and treatment of mental health disorders; home dialysis and end-stage renal disease-related visits; and diagnosis, evaluation, and treatment of acute stroke symptoms.
Manufacturer Refunds for Discarded Single-Use Vial Amounts
Drugs and biologicals are administered to patients in varying amounts; often, the amount administered is less than the total amount available in the drug’s vial or package. Some of these drugs are only available in single-dose vials or single-dose packages. The U.S. Food and Drug Administration (FDA)-approved labeling for a drug packaged in a single-dose container typically states that any extra amount of the drug remaining after a dose is administered must be discarded. Based on this language, under Part B, Medicare previously established that any unused and/or discarded amounts from a single-dose vial or single-dose package would be paid when reported on the claim with use of modifier JW. Note: modifier JW cannot be used with drugs that are not separately paid, such as drugs packaged into outpatient hospital services or other designated settings.
Section 90004 of the Infrastructure Investment and Jobs Act requires manufacturers to provide a refund to CMS for certain discarded amounts of a refundable single-dose container or single-use package drug. The refund amount is the amount of the discarded drug that exceeds an applicable percentage, which is required to be at least 10 percent, of the total charges for the drug in a given calendar quarter. CMS clarified that refundable single-dose vials or single-dose packages do not include radiopharmaceuticals, imaging agents, certain drugs requiring filtration, and certain new drugs. To accomplish the requirements of the Act, CMS proposed the following:
- Use modifier JW (or if another modifier is used or added in the future for the same data) to identify discarded billing units of a billing and payment code to calculate the refund amount.
- For dates of service on or after January 1, 2023, modifier JW will be required on claims for all single-dose container or single-use drugs when any amount is discarded, as part of CMS’ current policy.
- Use modifier JZ on billing claims to attest there was no discarded amount from the single-dose vial or single-use package that is normally paid under Part B.
- The definition for refundable single-dose container or single-use package drug would apply “to drugs paid under Medicare Part B (that is, under any payment methodology) that are described as being supplied in a ‘single-dose’ container or ‘single-use’ package based on FDA-approved labeling or product information. This definition also includes drugs described in FDA-approved labeling as a ‘kit’ that is intended for a single dose or single use.”
- Excluded drugs would be radiopharmaceuticals, imaging agents, drugs requiring filtration during the preparation process, and drugs approved on or after the Act’s date of enactment (November 15, 2021), for which payment under Part B has been made for fewer than 18 months.
- Exclusion of drugs requiring filtration during their preparation process specifically pertains to those in which the dosing and administration instructions that are included in the labeling require filtration during the drug preparation process, prior to dilution and administration, and that require any unused portion be discarded after the filtration process be discarded.
- Annual reports would be sent to drug manufacturers no later than October 1 of each year that include data from second, third, and fourth quarters of the previous year and the first quarter of the current year.
- Refunds by drug manufacturers would be due no later than December 31 of the year in which the annual report was delivered.
- Establishment of a dispute resolution process, civil monetary penalties, and periodic review of Part B medication claims to ensure modifier JW, modifier JZ (if finalized), and discarded drug amounts are billed appropriately as part of the already developed claims audit policy and process.
Comment Period
Comments for the MPFS proposed rule is open to stakeholders and must refer to file code CMS-1770-P. All comments must also be received no later than 5:00 PM EST on September 6, 2022. Electronic submission is encouraged by CMS. To submit your comments, follow the instructions under the “submit a comment” tab.
The CY 2023 MPFS final rule is expected to be released on or before November 1, 2022. This is when we will find out if the various payment policies and regulatory updates were finalized as proposed or something different.
Read my highlights from the CY2023 Hospital Outpatient Prospective Payment System proposed rule next.
Teri Bedard, BA, RT(R)(T), CPC, is executive director, Client & Corporate Resources at Revenue Cycle Coding Strategies in Des Moines, Iowa.