By Christian G. Downs, JD, MHA
On August 4, the Centers for Medicare & Medicaid Services (CMS) released its 2021 Medicare Physician Fee Schedule (PFS) and Hospital Outpatient Prospective Payment System (OPPS) proposed rules. The agency also released an accompanying executive order proposing increased flexibility for telehealth and rural healthcare in light of the COVID-19 pandemic.
CMS is waiving the 60-day publication requirement for the final rule and replacing it with a 30-day notification. The final rule will become effective January 1, 2021, although it may not be published until December 1, 2020. Comments on the proposed rule are due October 5, 2020.
OPPS Highlights
The OPPS proposed rule would continue many of the controversial policies CMS has implemented in recent years that have been upheld by the courts. These include the payment reduction for clinic visits at excepted (grandfathered) off-campus departments and the reduction in payment for drugs purchased under the 340B program. Overall, hospitals would see a 2.6% increase in payments under the proposed OPPS rule. This update is based on the projected hospital market basket increase of 3.0% minus a 0.4% adjustment for multi-factor productivity.
340B Program
CMS proposes increasing the 340B payment cut to ASP-28.7% from ASP-22.5%. The agency arrived at this number after concluding that survey data found an average acquisition cost of ASP-34.7%. CMS proposes to use ASP-34.7% as acquisition cost and add 6% of ASP for overhead and handling costs. CMS seeks comments on whether it should continue to pay for these drugs at ASP-22.5%. This policy has been the subject of ongoing litigation, and it was most recently upheld by the D.C. Circuit Court in July 2020.
CMS proposes continuing to reimburse drugs not purchased under the 340B program at ASP+6% if they have pass-through status or qualify for separate payment. The packaging threshold would remain at $130 for drugs without pass-through status.
Scope of Practice
CMS proposes to make permanent a policy finalized under the May 1 COVID-19 interim final rule that allows nurse practitioners, clinical nurse specialists, physician assistants, and certified nurse-midwives to supervise the performance of non-surgical extended duration therapy services (e.g., lengthy drug administration). Under current rules, these services require direct supervision (supervising practitioner is in the building and immediately available to assist) during the initiation of the service and general supervision for the rest of the service.
MAAAs
CMS proposes excluding certain cancer-related, protein-based multianalyte assays with algorithmic analyses (MAAAs) from the OPPS packaging policy and the date of service rule (14-day rule). This would allow these tests to be separately reimbursed under the Clinical Laboratory Fee Schedule.
PFS Highlights
Since the COVID-19 public health emergency (PHE) was declared earlier this year, CMS has issued waivers to increase flexibility and reduce regulatory burdens. In the PFS proposed rule, CMS proposes to make permanent, extend, or transition out of these new rules. ASCO notes that CMS estimates a plus-14% overall impact for hematology/oncology and a negative-6% impact for radiation oncology in 2021. The proposed CY 2021 PFS conversion factor is $32.26, a decrease of $3.83 from the CY 2020 MPFS conversion factor of $36.09.
E&M Visits
Per the CY 2020 MPFS final rule, beginning in 2021, CMS will largely align its E&M visit coding and documentation policies with changes enacted by the CPT® Editorial Panel for office/outpatient E&M visits. CMS proposes to clarify the times for which prolonged office/outpatient E&M visits can be reported and proposes revising the times used for rate-setting for this code set.
Quality Payment Program
Due to the COVID-19 pandemic, CMS will not introduce any Merit-based Incentive Payment System (MIPS) Value Pathways (MVP) for the 2021 performance period. CMS proposes a new alternative payment model (APM) performance pathway reporting option in 2021 to align with the MVP framework.
As part of the introduction of the performance pathway, CMS will sunset the CMS Web Interface as a collection type beginning in the 2021 performance period. CMS will continue to allow clinicians eligible for MIPS to participate in the program either as individuals or as part of a group or virtual group.
CMS is expanding the use of the APM entity submitter types to allow the use of all MIPS submission mechanisms. CMS proposes using performance period (rather than historical) benchmarks to score quality measures in 2021. The agency is concerned that it may not have a representative sample of historic data for 2019 (due to the pandemic), which would impact 2020 data submission, skewing benchmarking results.
Telehealth/Virtual Care
CMS proposes: 1) adding services to the Medicare telehealth list on a Category 1 basis, and 2) creating a third temporary category of criteria for services added to the Medicare telehealth list. “Category 3” will describe services added to the Medicare telehealth list during the COVID-19 PHE that will remain on the list through the calendar year in which the PHE ends.
In March 2020, CMS established separate payments for audio-only telephone E&M services. Although the agency is proposing to not continue to recognize these codes after the PHE ends, the agency states that it recognizes that “the need for audio-only interactions could remain as beneficiaries continue to try to avoid sources of potential infection, such as a doctor’s office.” Therefore, the agency is seeking comment on whether it should develop coding and payment for a service like the virtual check-in but for a longer unit of time and subsequently with a higher value. CMS is seeking comment on whether this service should be made permanent.
For the duration of the COVID-19 PHE, CMS has adopted an interim final policy revising the definition of direct supervision to include the virtual presence of the supervising practitioner using interactive audio/video real-time communications technology. CMS proposes continuing this policy through December 31, 2021.
Additional Resources
ACCC is reviewing these proposed rules and will provide comments by the Oct. 5 due date. To obtain further guidance from health policy experts about how these proposed rules may affect oncology practices, join our August 28 webcast at 3:00 pm EDT: CMS Proposed CY 2021 Payment Rules: What You Need to Know.
Christian G. Downs, JD, MHA, is executive director, Association of Cancer Care Centers.
The below is excerpted from the CMS fact sheet, Proposed Policy, Payment, and Quality Provisions Changes to the Medicare Physician Fee Schedule for Calendar Year 2021.
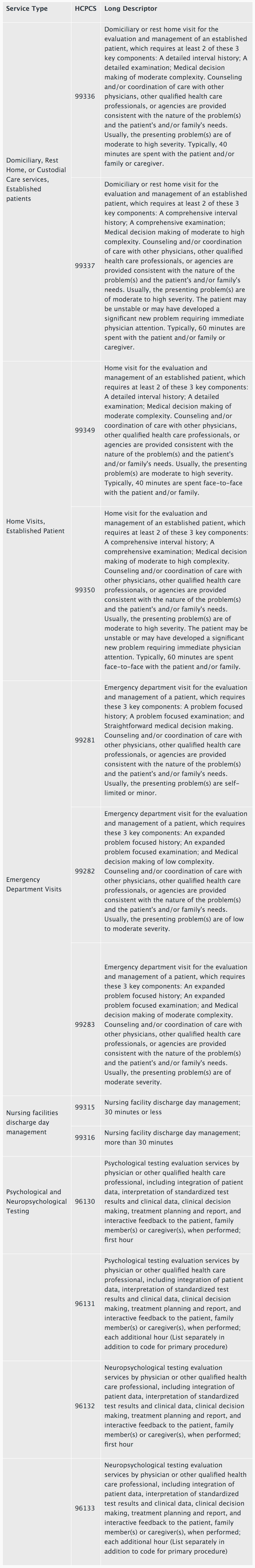
For CY 2021, CMS is proposing to add the following list of services to the Medicare telehealth list on a Category 1 basis. Services added to the Medicare telehealth list on a Category 1 basis are similar to services already on the telehealth list.
Additionally, we are proposing to create a third temporary category of criteria for adding services to the list of Medicare telehealth services. Category 3 describes services added to the Medicare telehealth list during the public health emergency (PHE) for the COVID-19 pandemic that will remain on the list through the calendar year in which the PHE ends.
We are proposing to add the following list of services to the Medicare telehealth list on a Category 3 basis: